Exela Pharma Sciences, LLC, (Exela) is voluntarily recalling the products listed in the table below to the consumer level. Particulate matter identified as silicone was observed during routine inspection of retain samples.
Risk Statement: Administration of an injectable product that contains particulate matter may result in local irritation or swelling in response to the foreign material. If the particulate matter reaches the blood vessels it can travel to various organs and block blood vessels in the heart, lungs or brain which can cause stroke and even lead to death. Exela has not received any reports of adverse events related to this recall.
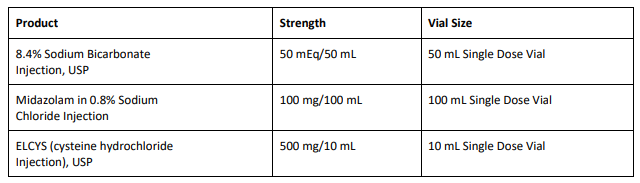
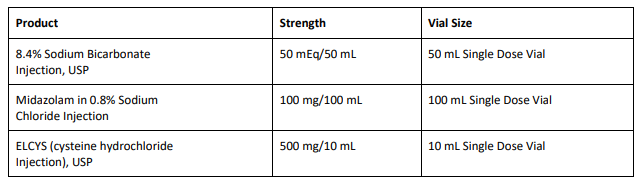
8.4% Sodium Bicarbonate Injection USP is used for treatment of metabolic acidosis and is packaged in a 50 mL glass single dose vials, 20 vials per carton Exela brand (Carton NDC: 51754-5001-5; Vial NDC: 51754-5001-1, Figure 1) and 25 vials per carton Exela brand (Carton NDC: 51754-5001-4; Vial NDC: 51754-5001-1) and Civica brand (Carton NDC: 72572-740-20; Vial NDC: 72572-740-01, Figure 2). The affected 8.4% Sodium Bicarbonate Injection, USP, 50 mEq/50 mL lots (covering both Exela and Civica brands) include the following lot numbers and expiration dates:
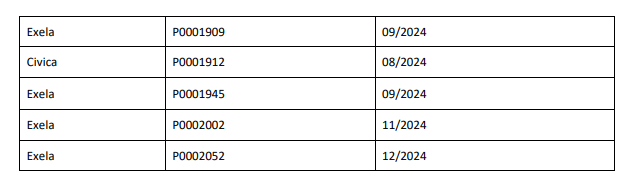
Product was distributed nationwide to wholesalers, distributors, and health systems between January 18,
2022, and February 15, 2023.


